Author links open overlay panel YasuteruSakuraiab, Mya MyatNgwe Tunc, YoheiKurosakiab, TakayaSakurad, Daniel KenInaokadf, KiyotakaFujinee, KiyoshiKitafg, KouichiMoritac, JiroYasudaab
- Department of Emerging Infectious Diseases, Institute of Tropical Medicine (NEKKEN), Nagasaki University, Nagasaki, 852-8523, Japan
- National Research Center for the Control and Prevention of Infectious Diseases, Nagasaki University, Nagasaki, 852-8521, Japan
- Department of Virology, Institute of Tropical Medicine (NEKKEN), Nagasaki University, Nagasaki, 852-8523, Japan
- Department of Molecular Infection Dynamics, Shionogi Global Infectious Diseases Division, Institute of Tropical Medicine (NEKKEN), Nagasaki University, Nagasaki, 852-8523, Japan
- Pharmaceutical Research Department, Global Pharmaceutical R&D Division, Neopharma Japan Co., Ltd, Tokyo, 102-0071, Japan
- School of Tropical Medicine and Global Health, Nagasaki University, Nagasaki, 852-8523, Japan
- Department of Host – Defense Biochemistry, Institute of Tropical Medicine (NEKKEN), Nagasaki University, Nagasaki, 852-8523, Japan
Highlights
- 5-amino levulinic acid (5-ALA) inhibited SARS-CoV-2 infection in cell culture.
- Antiviral activity of 5-ALA was more potent in human colon-derived Caco-2 cells than VeroE6 cells.
- The antiviral activity of 5-ALA was dose-dependent without significant cytotoxicity.
Abstract
The current COVID-19 pandemic requires urgent development of effective therapeutics. 5-amino levulinic acid (5-ALA) is a naturally synthesized amino acid and has been used for multiple purposes including as an anticancer therapy and as a dietary supplement due to its high bioavailability. In this study, we demonstrated that 5-ALA treatment potently inhibited infection of SARS-CoV-2, a causative agent of COVID-19, in cell culture. The antiviral effects could be detected in both human and non-human cells, without significant cytotoxicity. Therefore, 5-ALA is worth to be further investigated as an antiviral drug candidate for COVID-19.
Graphical Abstract:
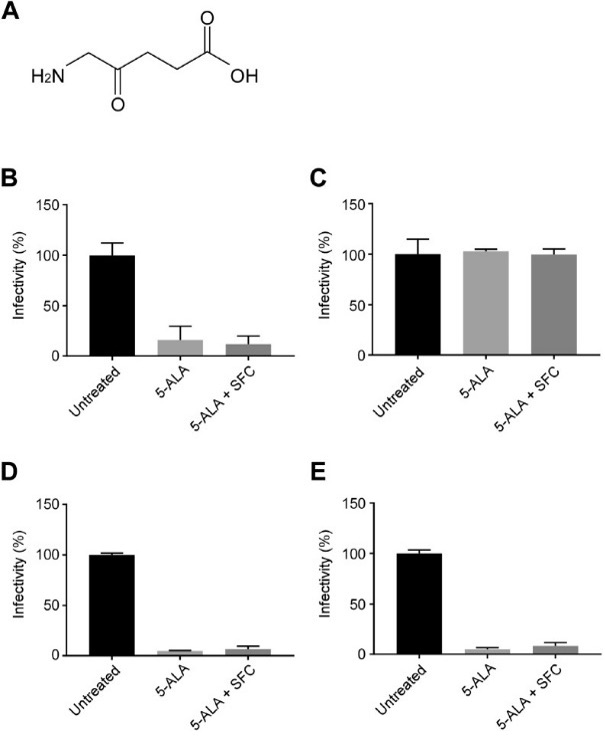
Fig. 2
5-ALA inhibits SARS-CoV-2 infection in both VeroE6 cells and Caco-2 cells. (A) A chemical structure of 5-ALA. (B and C) To address the effects of compounds on virus infection, VeroE6 cells were treated with 1000 μM of 5-ALA with and without 25 μM of SFC for 72 h (B) or 48 h (C) and challenged with SARS-CoV-2. Virus infectivity was calculated by counting the number of infected cells and normalizing it to untreated cells (mean ± SD, n = 3). (D and E) Infectivity of SARS-CoV-2 in Caco-2 cells with pretreatment for 72 h (D) or 48 h (E) were determined as (B) and (C). Each data set is representative of at least two independent experiments.
Copyright © 2021 Elsevier Inc. All rights reserved.
Journal Abstract: https://pubmed.ncbi.nlm.nih.gov/33571909/


No comment